Chemical Energetics
(4) Measuring the enthalpy change of reaction.
I
love sherbet that sweet tangy white stuff that used to come in a yellow tube
with a stick of liquorice or encased in rice paper or in a long straw or even
as a dib-dab!!.
Put
this stuff in your mouth and the thing fizzes and it goes cold.
What’s
going on?
Well
there is an endothermic reaction going on as the sherbet mixture dissolves in
your saliva.
As
it dissolves in your saliva your tongue feels cold: endothermic then….
And
the fizzing must be a gas given off.
That
all tells us that the reaction is a
neutralisation between a solid but soluble acid, in this case citric acid C6H8O7
, and a solid but soluble base, in this case sodium hydrogen carbonate NaHCO3
How
can we measure the enthalpy of reaction between citric acid and sodium
bicarbonate?
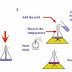
The
best approach to get at this enthalpy change is to use a polystyrene pot as in
the diagram below:
The
reaction takes place in solution and the temperature changes rising if the
reaction is exothermic and decreasing if the reaction is endothermic.
The
system is the reaction that takes place and the surroundings are the solution
in which the reaction happens.
We
can determine the energy transferred between reaction and the solution using
the simple formula
E = m
c ΔT
Where E is the energy transferred, m is the mass of solution c is the specific
heat capacity of the solution and ΔT the greatest temperature change during the
reaction.
If we use excess citric acid solution and add a known
mass of sodium hydrogen carbonate then we can determine the energy released per
mole of sodium hydrogen carbonate.
Here are a typical set of results and a calculation of
the enthalpy change:
Suppose we add 9.36g of sodium hydrogen carbonate
(0.111mol) to 31.64g of a 1.46M solution of citric acid.
The temperature change can be recorded every 15
seconds and the results plotted as a Temperature vs time graph.
The resulting plot looks like this:
You could also record this data using a temperature
probe and a datalogger.
From the graph you can determine the greatest
temperature fall.
This fall is 17oC down to 3oC a
fall of 14oC.
So using E
= m c ΔT
E = 31.64g *
4.18 J/g/oC * 14oC
E = 1852 Joules
In the polystyrene pot there were 0.111moles of sodium
bicarbonate and 0.046 mol citric acid.
This means that the citric acid is in excess.
3 moles sodium hydrogen carbonate react with one mole
citric acid
Here is the equation for the reaction:
3NaHCO3
+ H3C6H5O7 = Na3C6H5O7 +
3CO2 + 3H2O
Since 3*0.046 mol = 0.148 mol sodium hydrogen
carbonate are needed to react with 0.046 mol citric acid and only 0.111 mole
were added, it is the citric acid that is in excess and so it is the amount of
sodium hydrogen carbonate determines the values of the enthalpy change.
Therefore, 1852 Joules were taken in from the
surroundings when 0.111 mole of sodium hydrogen carbonate were
neutralized.
Scaling up to the value of energy released per mole of
sodium hydrogen carbonate
ΔH = 1852
J/mol = +16,684 kJ/mol
0.111














No comments:
Post a Comment